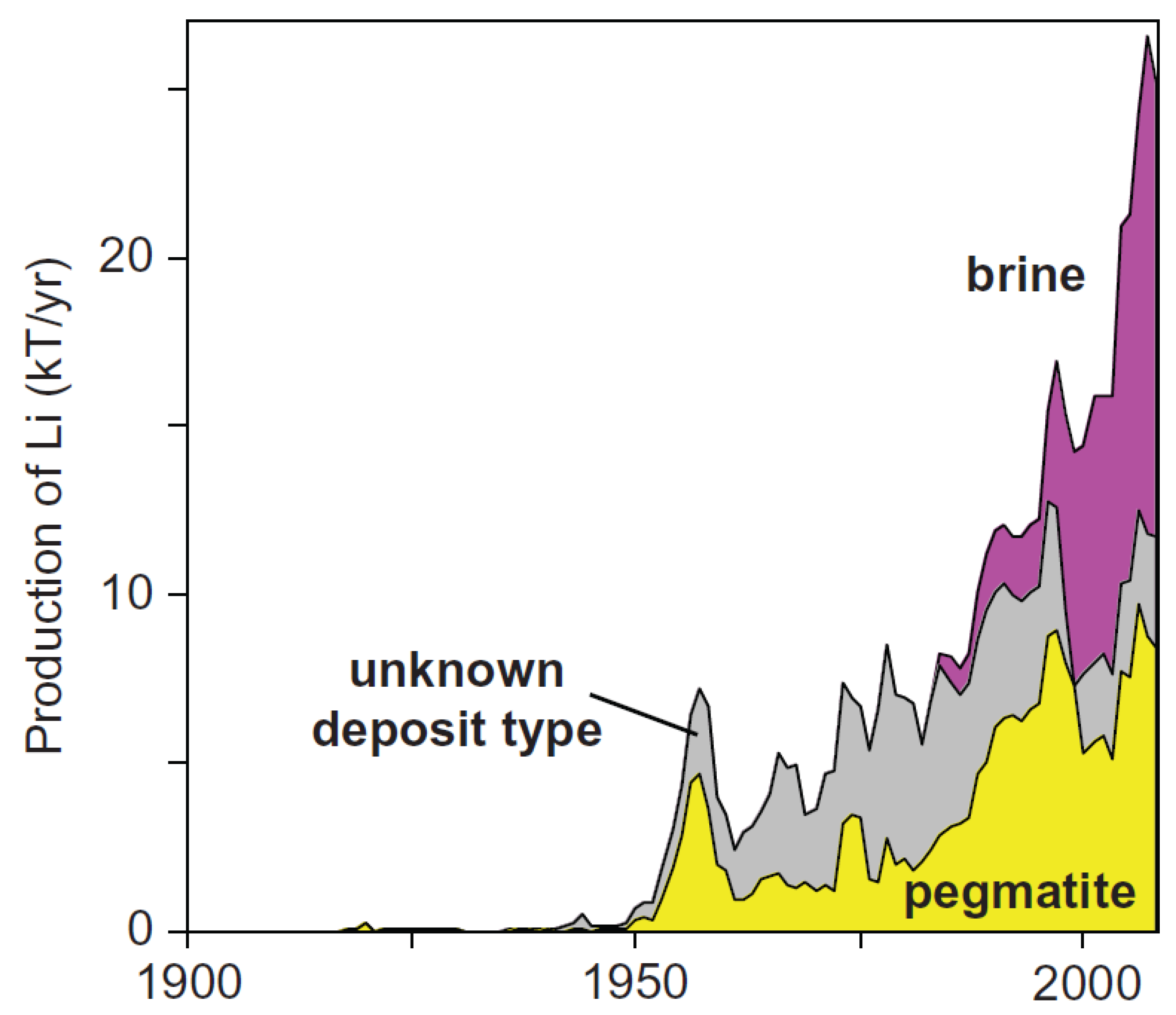
Engineering TiO2 supported CTAB modified bentonite for treatment of refinery wastewater through simultaneous photocatalytic oxidation and adsorption - ScienceDirect
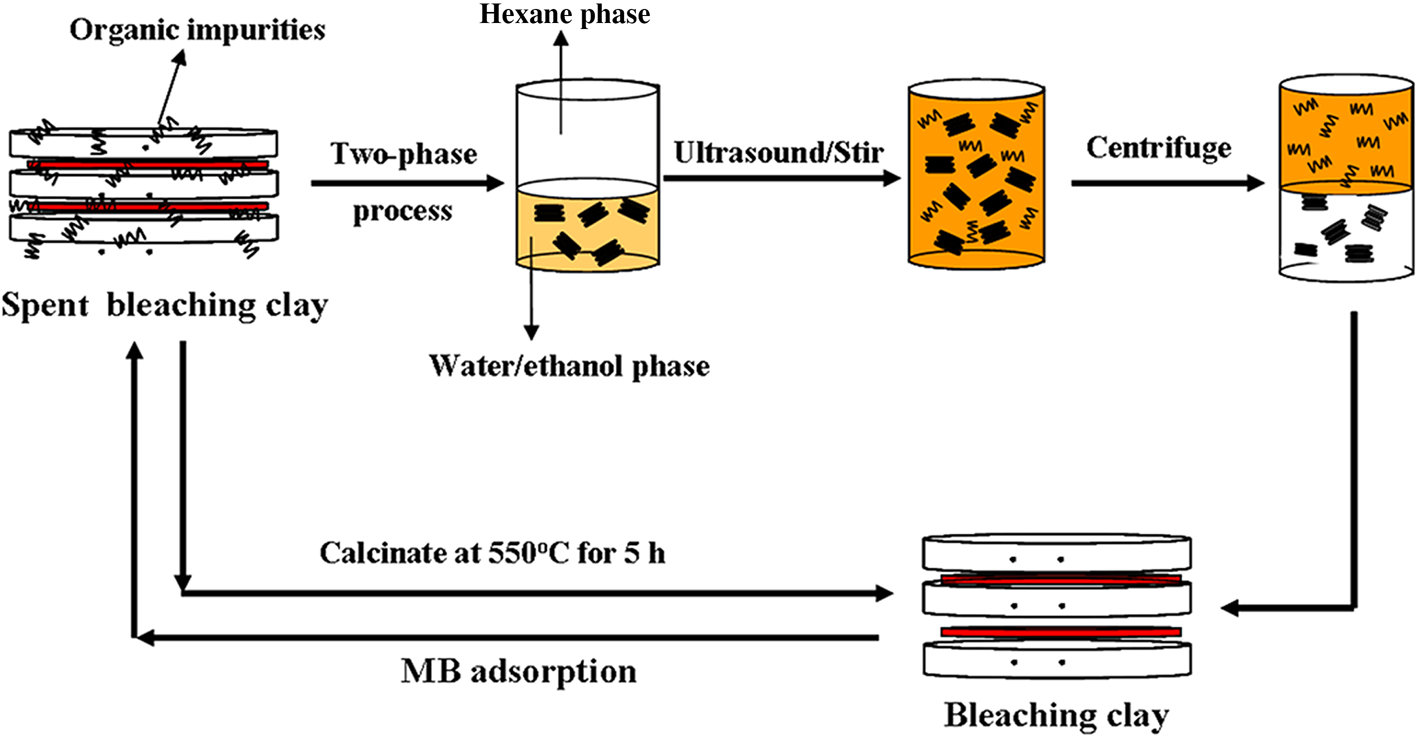
Regeneration of spent bleaching clay by ultrasonic irradiation and its application in methylene blue adsorption | Clay Minerals | Cambridge Core

PDF) Hydrogen Bonding and Vibrational Spectra in Kaolinite-Dimethylsulfoxide and -Dimethylselenoxide Intercalates–A Solid-State Computational Study

PDF) Hydrogen Bonding and Vibrational Spectra in Kaolinite-Dimethylsulfoxide and -Dimethylselenoxide Intercalates–A Solid-State Computational Study
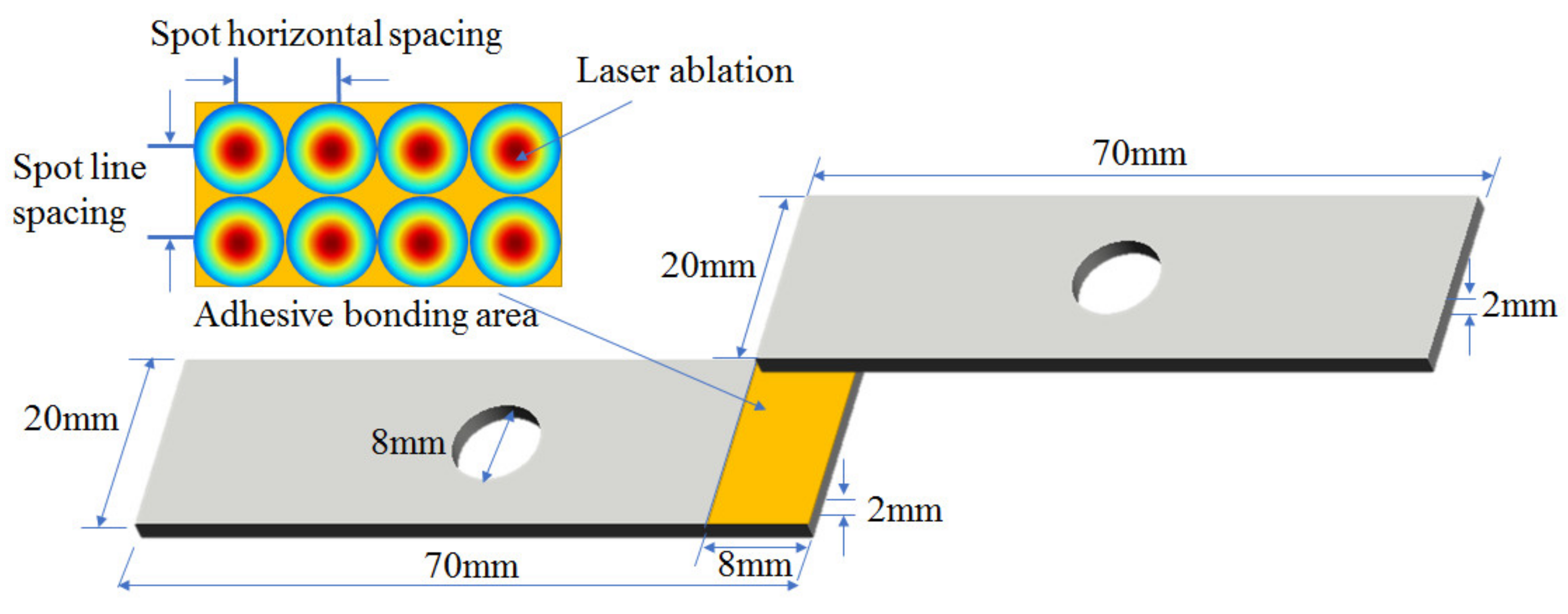
Applied Sciences | Free Full-Text | Improving Adhesive Bonding of Al Alloy by Laser-Induced Micro–Nano Structures | HTML
Potential energy of GPTMS molecule as a function of CCCO dihedral angle (). | Download Scientific Diagram
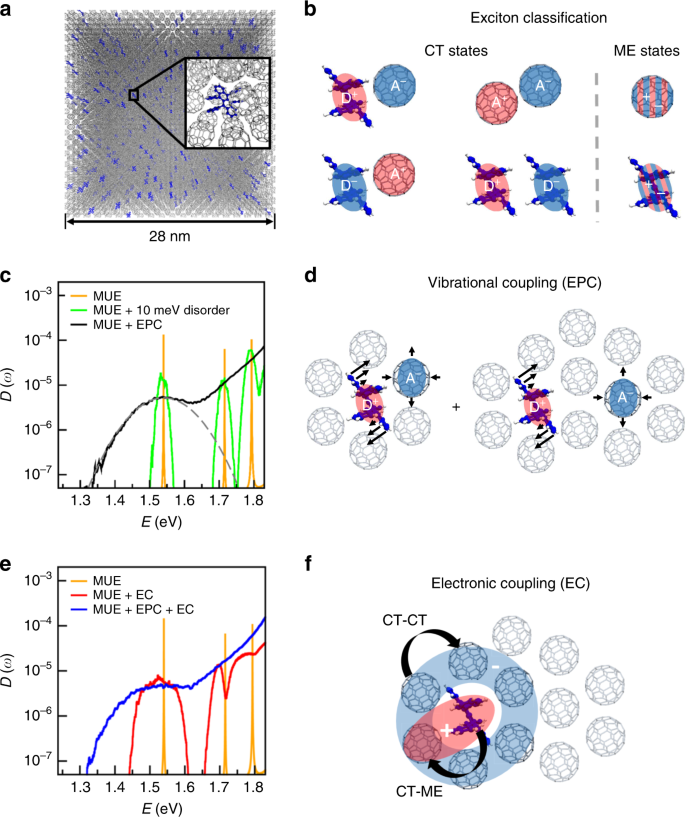
Molecular vibrations reduce the maximum achievable photovoltage in organic solar cells | Nature Communications
Potential energy of GPTMS molecule as a function of OCCO dihedral angle... | Download Scientific Diagram
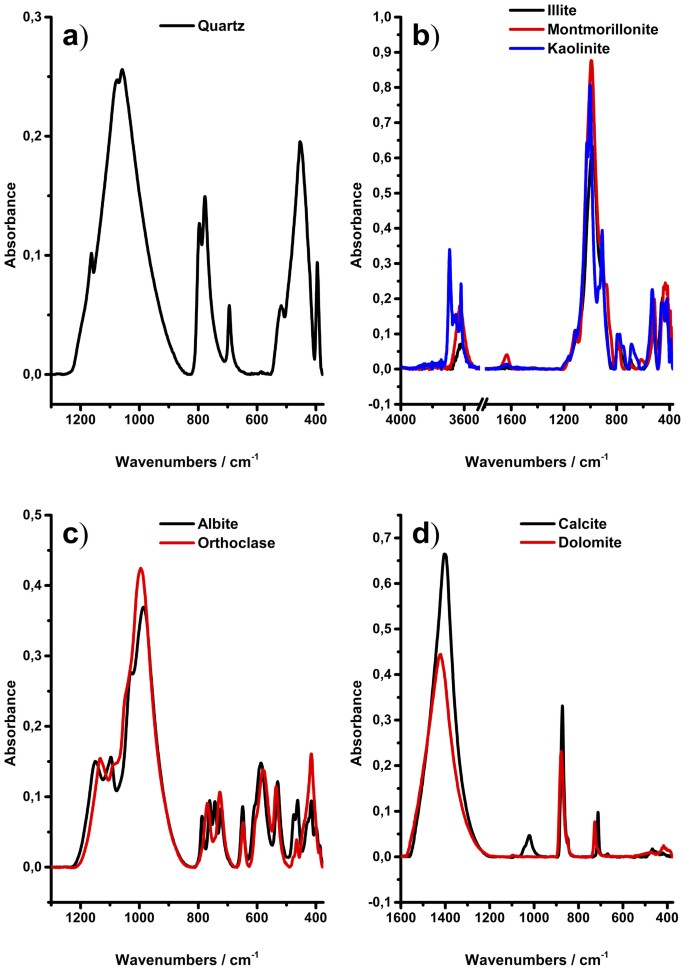
Infrared Attenuated Total Reflectance Spectroscopy: An Innovative Strategy for Analyzing Mineral Components in Energy Relevant Systems | Scientific Reports

Supramolecular Organization in Confined Nanospaces - Tabacchi - 2018 - ChemPhysChem - Wiley Online Library

Minerals | Free Full-Text | Distinguishing Features and Identification Criteria for K-Dioctahedral 1M Micas (Illite-Aluminoceladonite and Illite-Glauconite-Celadonite Series) from Middle-Infrared Spectroscopy Data | HTML

Fluid-Fluid Interactions Inducing Additional Oil Recovery during Low Salinity Water Injection in Inefficacious Presence of Clay Minerals - ScienceDirect
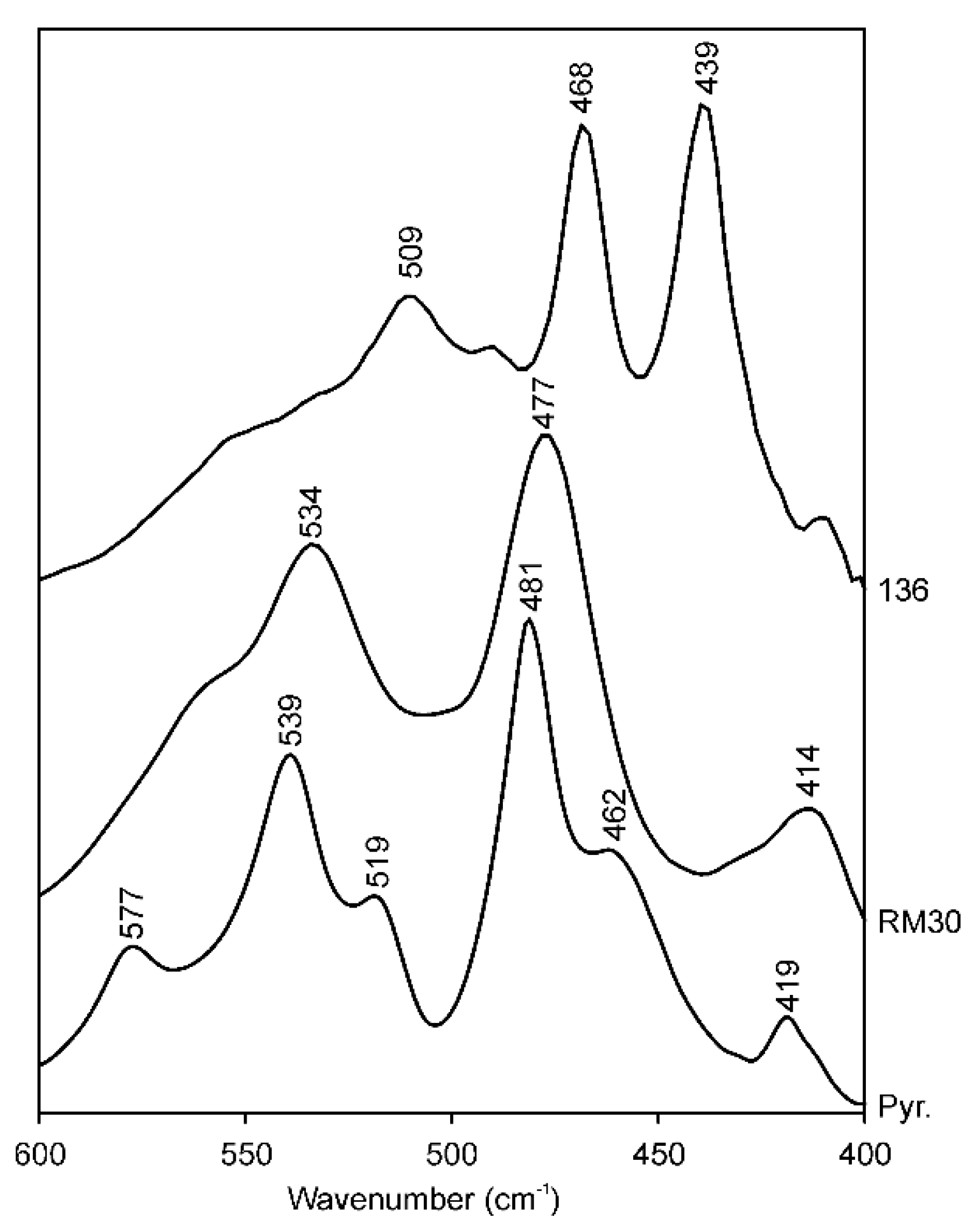
Minerals | Free Full-Text | Distinguishing Features and Identification Criteria for K-Dioctahedral 1M Micas (Illite-Aluminoceladonite and Illite-Glauconite-Celadonite Series) from Middle-Infrared Spectroscopy Data | HTML

Natural, low-cost adsorbents for toxic Pb(II) ion sequestration from (waste)water: A state-of-the-art review - ScienceDirect

Energies | Free Full-Text | Characteristics of Clay-Abundant Shale Formations: Use of CO2 for Production Enhancement | HTML

Synthesis and characterization of hybrid MgAl-LDH@SiO2@CoAl2O4 pigment with high NIR reflectance for sustainable energy saving applications - ScienceDirect
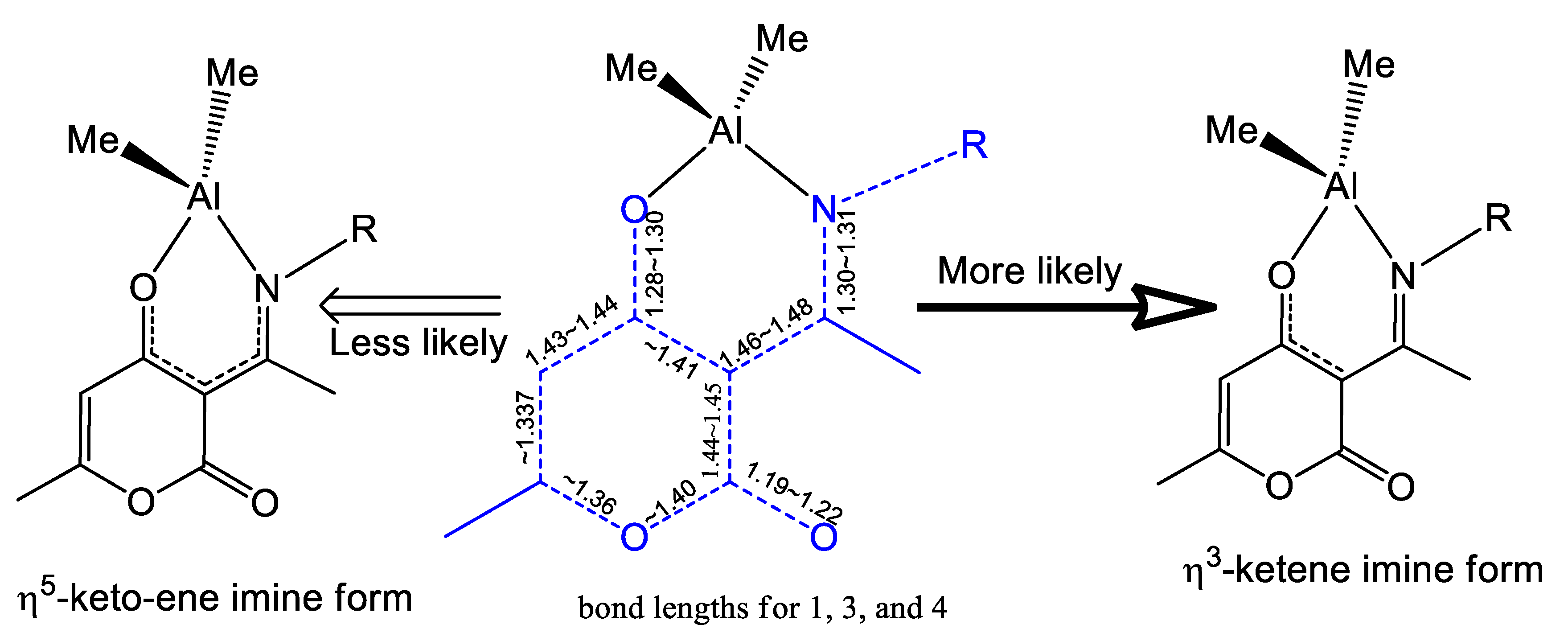
Molecules | Free Full-Text | Ring-Opening Polymerization of ε-Caprolactone and Styrene Oxide–CO2 Coupling Reactions Catalyzed by Chelated Dehydroacetic Acid–Imine Aluminum Complexes | HTML
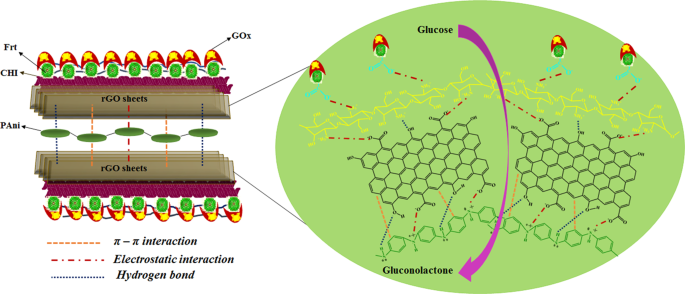
Applications of chitosan (CHI)-reduced graphene oxide (rGO)-polyaniline (PAni) conducting composite electrode for energy generation in glucose biofuel cell | Scientific Reports
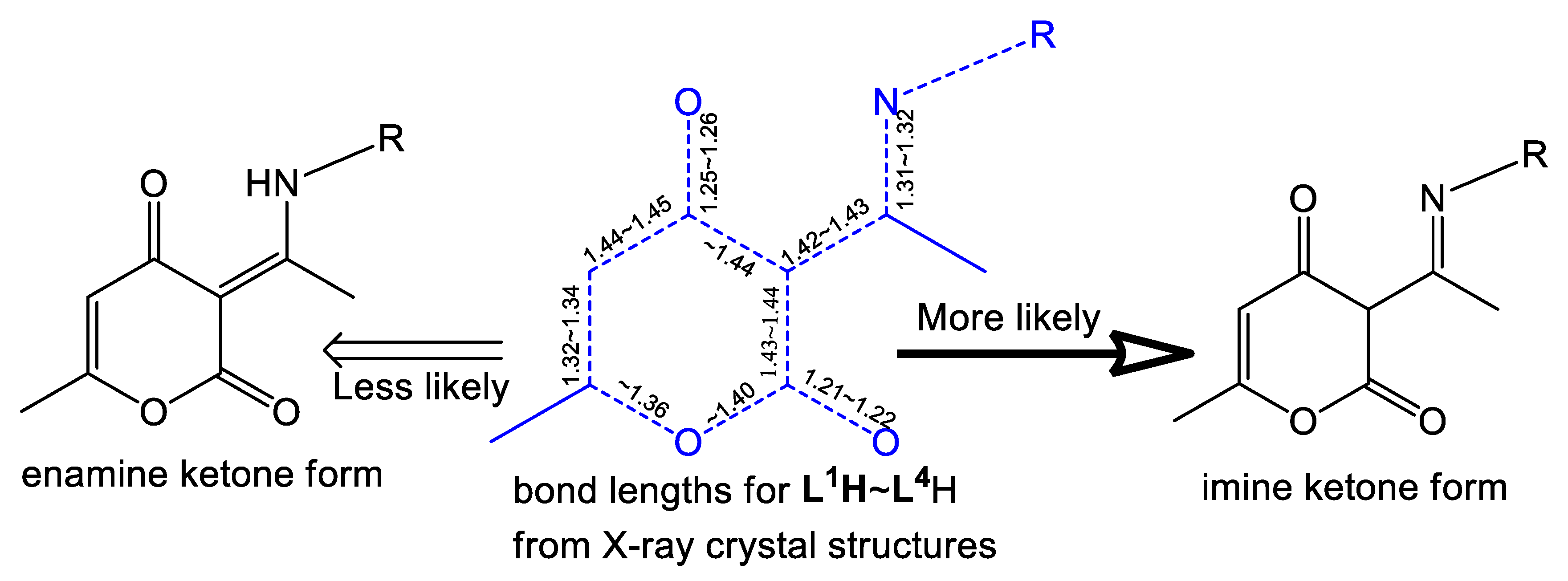
Molecules | Free Full-Text | Ring-Opening Polymerization of ε-Caprolactone and Styrene Oxide–CO2 Coupling Reactions Catalyzed by Chelated Dehydroacetic Acid–Imine Aluminum Complexes | HTML